Retatrutide and Cardarine: Exploring the Science Behind Combination Therapy
The pursuit of effective metabolic interventions has led researchers to explore increasingly sophisticated compound combinations. Among the most discussed pairings in the peptide and metabolic research community is Retatrutide — a triple-receptor agonist — combined with Cardarine (GW501516), a PPARδ agonist with a complex research history. This article examines both compounds, the scientific rationale for combining them, and the critical safety considerations that anyone following this research should understand.
What Is Retatrutide?
Retatrutide is an investigational peptide developed by Eli Lilly that acts as a triple agonist, simultaneously targeting three receptors: GLP-1 (glucagon-like peptide-1), GIP (glucose-dependent insulinotropic polypeptide), and glucagon receptors.
This triple-receptor mechanism sets it apart from earlier compounds like semaglutide (GLP-1 only) and tirzepatide (GLP-1/GIP dual agonist). Each receptor contributes a distinct metabolic effect:
- GLP-1 activation suppresses appetite through hypothalamic signaling, slows gastric emptying, and improves insulin sensitivity. Clinical trials have consistently demonstrated that GLP-1 agonists produce clinically meaningful reductions in HbA1c in type 2 diabetics.
- GIP activation enhances glucose-dependent insulin secretion and increases energy expenditure. Emerging research on dual-receptor agonists like tirzepatide has shown that combining GIP and GLP-1 receptor agonism produces substantially greater weight loss than GLP-1 agonism alone.
- Glucagon receptor activation promotes fat mobilization and hepatic energy expenditure. The Phase 2 trial published in the New England Journal of Medicine showed Retatrutide achieved up to 24.2% body weight reduction at the highest dose over 48 weeks (Jastreboff et al., NEJM 2023), with substantial preservation of lean mass.
Phase 2 trial data for Retatrutide showed up to 24% body weight reduction over 48 weeks. The TRIUMPH phase 3 program is currently underway, evaluating long-term efficacy and safety across obesity and type 2 diabetes endpoints.
What Is Cardarine (GW501516)?
Cardarine is a selective PPARδ (peroxisome proliferator-activated receptor delta) agonist — not technically a peptide, but frequently discussed alongside peptide protocols due to its metabolic effects. Originally developed in the early 1990s for dyslipidemia and cardiovascular disease, it targets a nuclear receptor that functions as a master regulator of cellular energy metabolism.
When PPARδ is activated, several downstream effects occur:
- Mitochondrial biogenesis — cells literally produce new mitochondria. Preclinical research has shown that PPARδ activation significantly increases mitochondrial density and biogenesis in skeletal muscle.
- Fuel substrate switching — the body shifts from glucose to fatty acid oxidation as its preferred energy source. Studies on PPARδ activation have demonstrated increased oxygen utilization with a metabolic shift toward preferential lipid oxidation.
- CPT-1 upregulation — carnitine palmitoyltransferase 1, the rate-limiting enzyme that shuttles fatty acids into mitochondria for oxidation, is significantly upregulated.
- Muscle fiber remodeling — PPARδ activation increases type I (slow-twitch oxidative) muscle fibers, which are inherently designed for fat burning and sustained energy production, as shown in 2006 PNAS research.
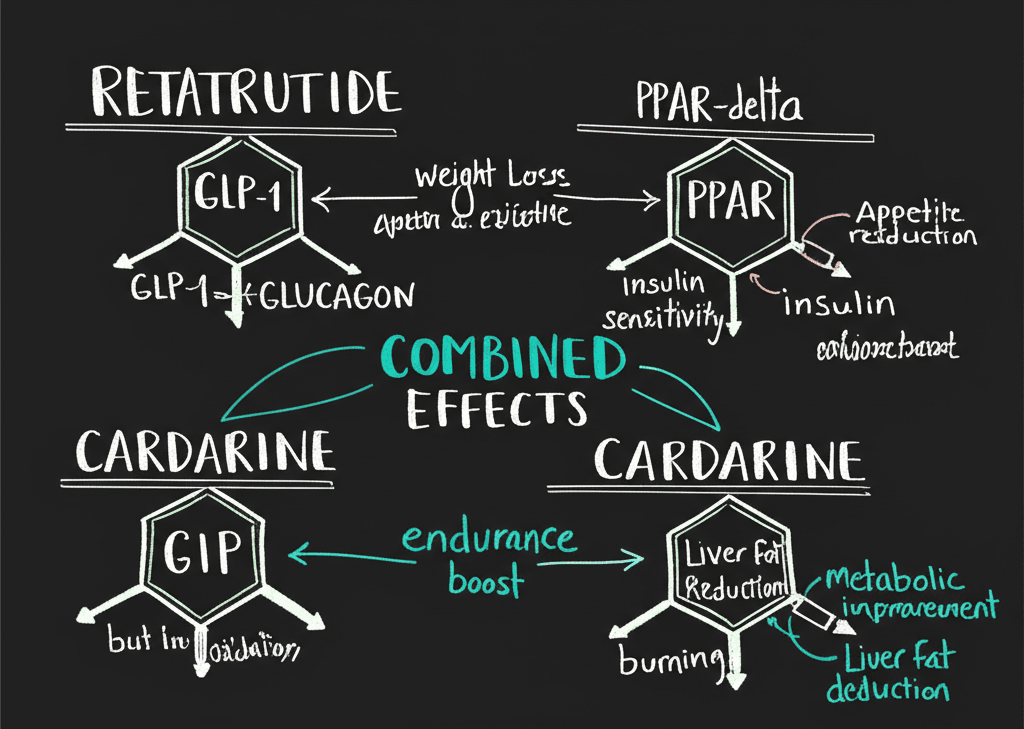
The Rationale for Combining Them
The theoretical basis for combining Retatrutide and Cardarine rests on their complementary mechanisms of action — one operates primarily through hormonal signaling (central regulation), while the other works at the cellular level (peripheral metabolism).
Directed Fat Loss With Muscle Preservation
Retatrutide creates a caloric deficit through appetite suppression and increased energy expenditure. However, caloric deficits typically trigger a metabolic tradeoff: the body may catabolize muscle tissue for fuel. This is a well-documented limitation of GLP-1 monotherapy, where lean mass loss can accompany fat loss.
Cardarine’s PPARδ activation preferentially directs energy metabolism toward fat oxidation while sparing muscle protein. Research suggests that PPARδ activation may preferentially direct energy metabolism toward fat oxidation while helping preserve lean muscle mass — essentially directing the body to burn fat stores rather than muscle.
The proposed synergy: Retatrutide creates the deficit, Cardarine directs that deficit toward fat stores rather than muscle.
Breaking Metabolic Plateaus
Weight loss plateaus occur when the body activates metabolic adaptation — sympathetic nervous system downregulation, reduced non-exercise activity thermogenesis (NEAT), and decreased thyroid hormone production. This is an evolutionary survival mechanism that makes sustained fat loss progressively harder.
Cardarine may counteract this by sustaining elevated basal metabolic rate through increased mitochondrial density and oxidative capacity. A landmark study demonstrated that PPARδ activation promotes fat metabolism and resistance to obesity through increased energy uncoupling (Wang et al., Cell 2003) — representing sustained metabolic reprogramming, not a temporary boost.
Retatrutide appears to prevent the hormonal component of metabolic adaptation. Retatrutide's glucagon receptor component is theorized to increase energy expenditure during caloric deficits, potentially counteracting the metabolic adaptation that typically slows weight loss.
Insulin Resistance and Metabolic Healing
Insulin resistance — when cells stop responding properly to insulin — is driven in part by lipotoxicity: the accumulation of lipids within muscle cells and hepatocytes that physically impairs insulin signaling cascades. Research has consistently shown that increasing fatty acid oxidation reduces intramuscular lipid accumulation and improves insulin sensitivity, independent of weight loss — a well-established concept in metabolic physiology.
Cardarine addresses the lipotoxic blockade by clearing intracellular lipid accumulation through enhanced fatty acid oxidation. Retatrutide then optimizes insulin secretion timing and pancreatic beta cell function while improving hepatic insulin sensitivity through glucagon receptor activation.
Phase 2 data showed Retatrutide monotherapy reduced fasting insulin by up to 70.9% and improved HOMA2-IR (insulin resistance index) by up to 69.3% at 48 weeks (Hartman et al., Nature Medicine 2024).
Hepatic Fat Reduction
Non-alcoholic fatty liver disease (NAFLD) affects approximately 30% of the population. The progression from fat accumulation to inflammation to fibrosis to cirrhosis represents a serious clinical concern.
PPARδ activation directly increases hepatic fatty acid oxidation. A 2017 study showed that the selective PPARδ agonist seladelpar significantly reduced hepatic triglycerides and reversed NASH pathology in preclinical models (Haczeyni et al., Hepatology Communications 2017). Retatrutide complements this by reducing the delivery of free fatty acids to the liver and decreasing hepatic de novo lipogenesis.
Retatrutide monotherapy data showed greater than 80% mean relative liver fat reduction at higher doses in patients with metabolic dysfunction-associated steatotic liver disease (Hartman et al., Nature Medicine 2024).
Safety Considerations: A Critical Assessment
While the mechanistic rationale is compelling, any discussion of this combination must address significant safety concerns — particularly regarding Cardarine.
Cardarine’s Cancer Risk
Cardarine’s development was halted by GlaxoSmithKline around 2007 after high-dose rodent studies showed rapid tumor development across multiple organs. This finding led to the abandonment of all human clinical trials. No completed long-term human safety data exists.
It is important to note that rodent cancer models do not always translate directly to humans, and the doses used in these studies were significantly higher than those typically discussed in research contexts. However, the absence of long-term human safety data means the actual cancer risk in humans remains unknown — and this uncertainty is itself a serious concern.
Cardarine is banned by WADA (World Anti-Doping Agency) and is not approved for human use in any jurisdiction.
Retatrutide’s Investigational Status
Retatrutide remains in phase 3 clinical trials and is not FDA-approved as of 2026. Common side effects observed in trials include nausea, diarrhea, vomiting, and decreased appetite — consistent with the GLP-1 agonist class. Long-term safety data is still being collected.
No Combination Studies Exist
Critically, no clinical trials have studied the Retatrutide-Cardarine combination in humans. All proposed synergistic effects are inferred from the individual compounds’ mechanisms of action and their separate bodies of research. The actual interaction profile — including potential adverse effects unique to the combination — is entirely unknown.
Regulatory Reality
Neither compound is available through standard medical channels for general use. Retatrutide is accessible only through clinical trials or specialized research contexts. Cardarine exists in a gray market as a “research chemical” — its sale for human consumption is illegal in many jurisdictions.
What the Research Suggests — and What It Doesn’t
The mechanistic case for combining Retatrutide and Cardarine is grounded in legitimate biochemistry:
- Complementary pathways: Central hormonal regulation (Retatrutide) paired with peripheral cellular reprogramming (Cardarine) addresses metabolic dysfunction from multiple angles simultaneously.
- Preclinical support: Individual studies on each compound’s mechanisms suggest non-overlapping benefits that could theoretically amplify each other.
- Metabolic syndrome relevance: Given that an estimated 88% of Americans exhibit some degree of metabolic dysfunction (per 2019 CDC data), interventions targeting root metabolic causes rather than symptoms represent an important research direction.
However, the research does not yet support:
- Specific dosing protocols for the combination
- Long-term safety of either compound used together
- Definitive clinical outcomes in human subjects using both compounds
- Any claims of disease “cure” or reversal without concurrent lifestyle modification
The Bottom Line
Retatrutide and Cardarine represent two of the most mechanistically interesting compounds in metabolic research. Their complementary pathways — Retatrutide’s triple-receptor hormonal optimization and Cardarine’s PPARδ-driven cellular reprogramming — create a theoretical framework for addressing fat loss, insulin resistance, metabolic adaptation, and hepatic steatosis simultaneously.
But theoretical frameworks and proven therapies are separated by clinical trials, safety data, and regulatory approval. The science behind each compound individually is compelling and supported by peer-reviewed research. The case for combining them is logical but unproven in controlled human studies.
Anyone following this research should:
The metabolic research landscape is advancing rapidly. These compounds — individually and potentially in combination — represent genuine scientific progress in understanding how to reprogram human metabolism. The key is to follow the science with both enthusiasm and the rigor it demands.
References
🛒 Shop Featured Peptides
Peptides mentioned in this article are available from Mile High Compounds. Use our referral link to support independent peptide research.
These are third-party products. This is not medical advice. Consult your physician before use. Affiliate links support this site.