TB-500 and Cardiac Fibrosis: What the Research Says About AFib and Tissue Repair
Atrial fibrillation (AFib) is the most common sustained cardiac arrhythmia, affecting an estimated 33 million people worldwide. At its core, AFib is driven by structural remodeling of the heart — particularly cardiac fibrosis, the excessive accumulation of extracellular matrix proteins that disrupts normal electrical conduction.
TB-500, a synthetic peptide derived from the active region of Thymosin Beta-4 (Tβ4), has attracted research interest for its potential to address the fibrotic processes underlying AFib and other cardiac conditions.
Understanding Cardiac Fibrosis
Cardiac fibrosis occurs when fibroblasts — the cells responsible for producing structural proteins like collagen — become overactive. This can happen after a heart attack, during chronic hypertension, or as part of age-related cardiac remodeling.
The result is a heart with patches of stiff, scarred tissue that:
- Disrupts electrical signaling — creating the erratic conduction pathways that characterize AFib
- Reduces contractile efficiency — the heart can't pump as effectively when scarred tissue replaces functional cardiomyocytes
- Promotes arrhythmia recurrence — even after cardioversion or ablation, fibrotic tissue can serve as a substrate for re-entry circuits
Thymosin Beta-4 and Cardiac Repair: The Preclinical Evidence
Thymosin Beta-4 (Tβ4), the parent protein of TB-500, has been the subject of multiple preclinical studies examining its effects on cardiac tissue.
Key Findings
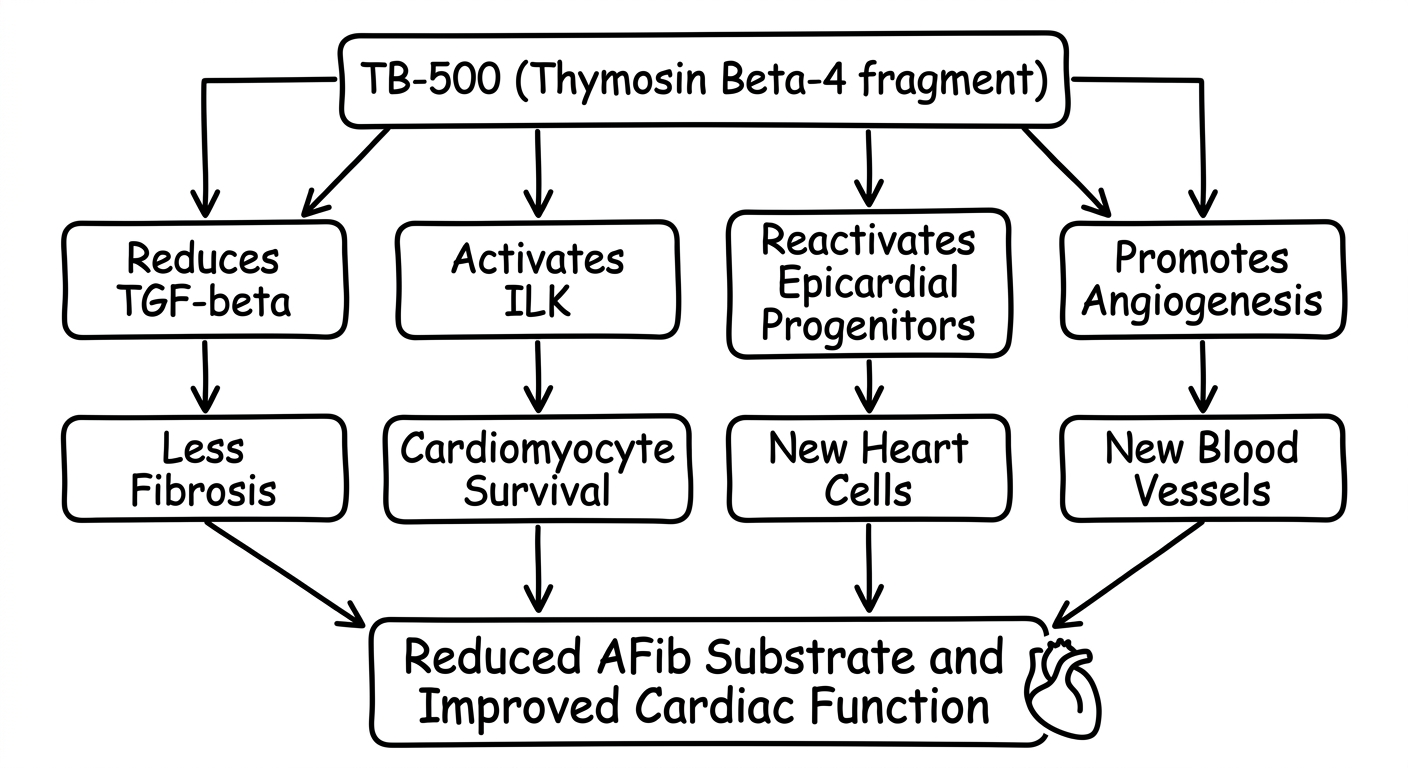
Anti-fibrotic effects: Studies in murine models of myocardial infarction have demonstrated that Tβ4 treatment significantly reduces collagen deposition and fibrotic scar size. The mechanism appears to involve downregulation of transforming growth factor beta (TGF-β), a master regulator of fibrosis, and modulation of matrix metalloproteinase (MMP) activity. Cardiomyocyte survival: Research published in the Proceedings of the National Academy of Sciences showed that Tβ4 activates integrin-linked kinase (ILK), which promotes cardiomyocyte survival following ischemic injury. Treated animals showed smaller infarct areas and improved left ventricular function compared to controls. Epicardial progenitor activation: One of the most intriguing findings is that Tβ4 can reactivate epicardial progenitor cells in adult hearts. These cells, normally quiescent after embryonic development, can differentiate into new cardiomyocytes and vascular smooth muscle cells — essentially enabling a limited form of cardiac regeneration. Angiogenesis: Tβ4 strongly promotes new blood vessel formation in ischemic cardiac tissue. By upregulating actin polymerization and promoting endothelial cell migration, it helps restore blood supply to damaged areas of the heart.TB-500 vs. Thymosin Beta-4: An Important Distinction
TB-500 is not identical to full-length Thymosin Beta-4. It is a synthetic peptide corresponding to the active region (amino acids 17–23) of the Tβ4 sequence. While TB-500 retains the actin-binding and cell migration properties of the parent protein, the extent to which it replicates all of Tβ4's cardiac benefits remains an active area of investigation.
Most published cardiac research has used full-length Tβ4 rather than the TB-500 fragment. Extrapolating these results directly to TB-500 requires caution, though the shared mechanism of action (actin regulation, cell migration, angiogenesis) provides a reasonable biological rationale.
The AFib Connection
The link between TB-500/Tβ4 and atrial fibrillation is primarily through the anti-fibrotic pathway:
Animal models of atrial fibrosis have shown that Tβ4 treatment reduces atrial collagen content and decreases the inducibility of atrial arrhythmias. However, no human clinical trials have specifically tested TB-500 or Tβ4 for AFib treatment.
Current Limitations
- Preclinical data only: The cardiac benefits of Tβ4/TB-500 have been demonstrated primarily in rodent models. Human cardiac physiology differs in important ways, and these results may not translate directly.
- Dosing uncertainty: Optimal dosing for cardiac applications is unknown. Most preclinical studies used direct intracardiac or intraperitoneal delivery — very different from the subcutaneous injections typically used with TB-500.
- Long-term safety: The effects of chronic TB-500 use on cardiac tissue are unstudied. Theoretical concerns about promoting unwanted cell proliferation have not been resolved.
- Regulatory status: TB-500 is not FDA-approved for any cardiac indication. It remains a research compound.
What This Means for Peptide Research
The Tβ4/TB-500 cardiac fibrosis research represents one of the most promising frontiers in peptide-based cardiovascular therapy. The ability to potentially reduce fibrotic remodeling, promote angiogenesis, and activate endogenous cardiac repair mechanisms addresses the root causes of conditions like AFib — not just the symptoms.
However, the gap between promising preclinical results and proven human therapies is significant. Anyone interested in this area should:
The Bottom Line
TB-500 and its parent protein Thymosin Beta-4 show genuine scientific promise for addressing cardiac fibrosis — the structural driver behind AFib and other cardiac conditions. The anti-fibrotic, pro-angiogenic, and regenerative properties observed in animal models are compelling. But we are still in the early stages of understanding how these findings translate to human cardiac care. The research deserves attention, and the science is advancing — but patience and rigor are essential as the field moves toward clinical validation.
🛒 Shop Featured Peptides
Peptides mentioned in this article are available from Mile High Compounds. Use our referral link to support independent peptide research.
These are third-party products. This is not medical advice. Consult your physician before use. Affiliate links support this site.