Why Cold Sores Keep Coming Back and preventing it
Why Cold Sores Keep Coming Back: The Neuroscience Behind HSV-1 Reactivation and Research-Backed Prevention Protocols
If you have ever suffered through recurring cold sores and wondered why antiviral medications seem to offer diminishing returns over time, you are not alone — and the answer lies far deeper than your skin. This article breaks down the precise neurological mechanisms that allow herpes simplex virus type 1 (HSV-1) to evade your immune system indefinitely, explains every biological trigger that drives reactivation, and outlines the most evidence-supported compounds, peptides, and lifestyle interventions that research associates with dramatically reducing outbreak frequency.
Herpes Is Not a Skin Condition — It Is a Neurological Hijacker
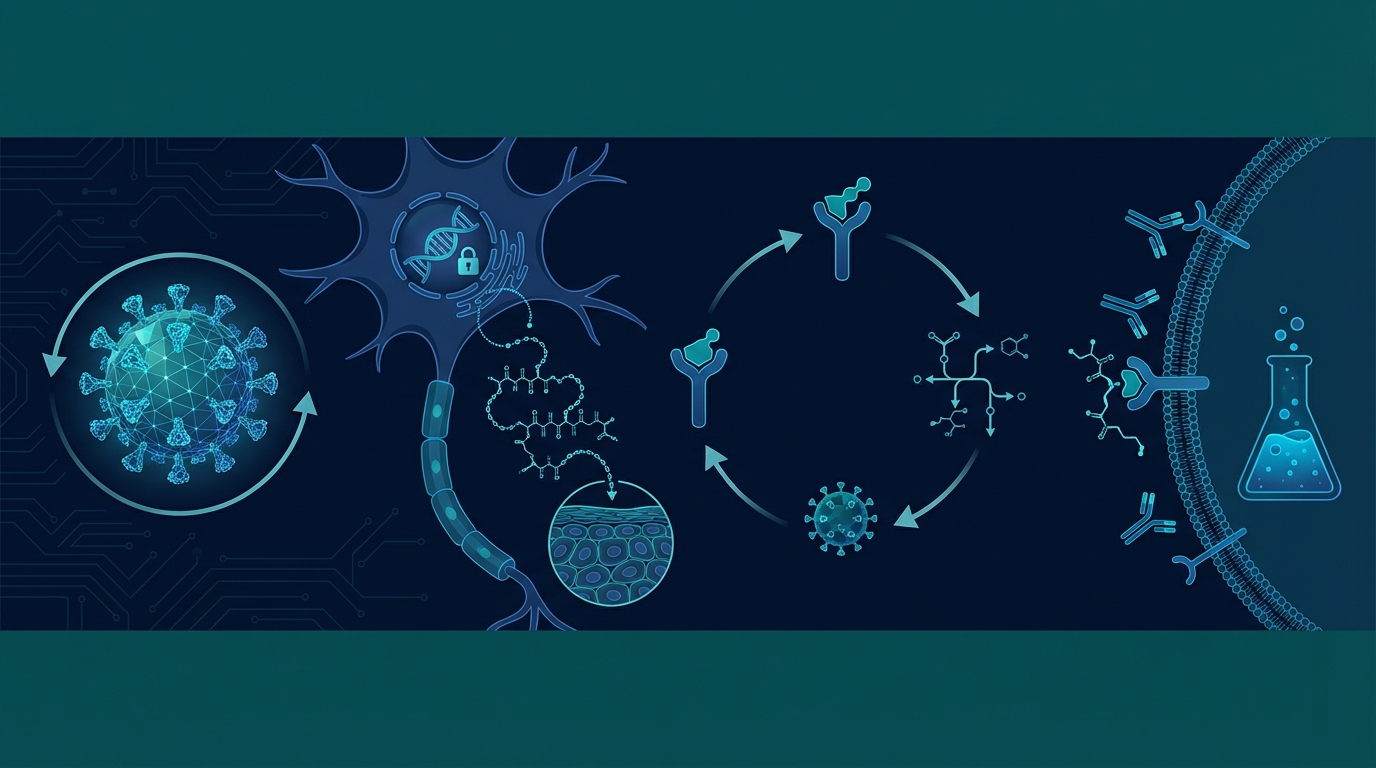
One of the most persistent and damaging misconceptions in popular medicine is that cold sores are primarily a skin infection. They are not. HSV-1 uses your skin merely as an exit ramp. The true battlefield is your nervous system.
When the herpes simplex virus first enters the body — typically through a break in the skin or mucous membranes — it does not stay at the site of entry. Within hours, the virus actively seeks out sensory nerve endings: the fine, branching fibers responsible for detecting pain, temperature, and touch. These nerve terminals extend from the surface of your skin all the way back to nerve cell clusters called ganglia.
For oral herpes (HSV-1), the destination is the trigeminal ganglion, a cluster of nerve cell bodies located at the base of the skull. For genital herpes (HSV-2), the virus migrates to the sacral ganglion in the lower spine. Understanding this distinction is fundamental, because it explains why topical creams applied to the lip blister accomplish almost nothing once an outbreak is underway — you are treating the exhaust pipe, not the engine.

How HSV-1 Travels Up Your Axon and Establishes Latency
The mechanics of how HSV-1 reaches the ganglion involve a process that is elegant and deeply frustrating in equal measure. Research published in Current Opinion in Virology (Heldwein & Krummenacher, 2008, with subsequent work extending into 2015) confirmed that herpes simplex virus exploits microtubule-associated dynein motors — protein machines inside nerve cells that normally transport essential cargo along the axon toward the cell body.
In plain terms: the virus disguises itself as routine cellular freight. Dynein motors physically carry viral DNA and proteins up the long axon — sometimes a journey of several centimeters — all the way to the nerve cell body inside the ganglion. Once there, the viral DNA is released directly into the cell's nucleus.
Here is where the biology becomes particularly challenging. Unlike many pathogens that immediately begin replicating and killing host cells, HSV-1 does the opposite. It enters a state called latency. The virus ceases replication entirely. It does not produce viral proteins that immune cells can recognize. It simply exists, silently integrated into the genetic machinery of your neuron.
This is why the immune system — specifically T-cells — cannot eliminate herpes without destroying the very sensory neurons in which it resides. Any therapeutic approach that claims to fully "cure" herpes would, by definition, require eradicating infected neurons, which would devastate the sensory nervous system. This biological reality is not a marketing disclaimer; it is hard immunology.
The Three Biological Triggers That Reactivate Dormant HSV-1
Latent HSV-1 does not stay dormant forever. Reactivation — the journey back down the axon toward the skin surface, where new viral particles cause blistering — is driven by specific, identifiable biological failures. Research consistently points to three interrelated systemic dysfunctions:
- Systemic Inflammation: Elevated pro-inflammatory cytokines, particularly IL-6, TNF-alpha, and IL-1β, create a cellular environment that destabilizes latency. Chronic low-grade inflammation is now understood to be one of the most reliable predictors of outbreak frequency.
- Insulin Resistance and Metabolic Dysregulation: High circulating glucose and impaired insulin signaling suppress immune surveillance. NK (natural killer) cells and CD8+ T-cells — the primary defenders against HSV reactivation — are significantly less effective in hyperglycemic environments. Poor blood sugar regulation directly correlates with increased outbreak frequency.
- Impaired Mitochondrial ATP Production: Nerve cells are among the most energetically demanding cells in the human body. When mitochondrial function declines — due to nutrient deficiency, chronic stress, or oxidative damage — neurons cannot mount adequate antiviral defense. ATP depletion in the trigeminal ganglion is strongly associated with viral reactivation events.
A recognizable outbreak pattern often emerges: a period of elevated stress leads to poor sleep, poor sleep drives systemic inflammation and cortisol dysregulation, cortisol suppresses immune function, and the energy-depleted nerve cell can no longer suppress the latent virus. The prodromal tingling begins, and the outbreak follows predictably within days.
Why Long-Term Antiviral Medications Often Fail — and Their Hidden Costs
Acyclovir, valacyclovir, and famciclovir are the three antiviral medications most commonly prescribed for HSV suppression. These drugs work by inhibiting viral DNA polymerase — essentially blocking viral replication during an active outbreak or, at suppressive doses, attempting to prevent reactivation altogether.
The problem is structural. These antivirals do not address the latent reservoir in the ganglion. They cannot reach the dormant viral DNA in a non-replicating neuron. When the three core biological triggers listed above are not corrected — inflammation, insulin resistance, and ATP insufficiency — the immune environment that permits reactivation remains intact, and outbreaks continue despite medication.
Furthermore, long-term antiviral use carries renal considerations that are rarely discussed. Both acyclovir and valacyclovir are nephrotoxic at elevated doses and require adequate hydration and periodic kidney function monitoring during extended use. Years of continuous suppressive antiviral therapy without addressing underlying metabolic health creates a situation where the patient becomes dependent on a pharmaceutical that treats only the symptom of a deeper systemic problem.
Research-Backed Compounds and Peptides for HSV-1 Outbreak Reduction
Addressing the three root drivers — inflammation, insulin resistance, and mitochondrial ATP production — requires a multi-pronged approach. The following compounds have meaningful research supporting their relevance to either HSV suppression or the biological pathways that govern it.
L-Lysine
L-Lysine is the most extensively studied amino acid in the context of HSV suppression. Its mechanism involves competitive antagonism with L-arginine: the herpes virus requires arginine as a structural building block for viral replication. Lysine competes with arginine for intestinal absorption and cellular uptake, effectively starving the virus of a key replication resource. Clinical trials, including a randomized controlled study published in Dermatologica, demonstrated that lysine supplementation at doses of 1,000–3,000 mg per day significantly reduced outbreak frequency and severity compared to placebo. Dietary arginine restriction (reducing nuts, seeds, chocolate, and certain grains) combined with lysine supplementation represents a well-validated foundational intervention.
Zinc (Zinc Sulfate / Zinc Gluconate)
Zinc has demonstrated direct antiviral activity against HSV-1 in both in vitro and clinical studies. Zinc ions interfere with viral capsid protein assembly and inhibit HSV-1 DNA replication. Topical zinc sulfate (0.025–0.05% solution) has shown efficacy in reducing healing time when applied at prodrome. Systemic zinc supplementation at 25–45 mg elemental zinc daily supports NK cell function and CD8+ T-cell activity — both critical for maintaining HSV latency. Zinc deficiency is strongly correlated with increased reactivation events.
BPC-157 (Body Protection Compound-157)
BPC-157 is a synthetic pentadecapeptide derived from a protective protein found in gastric juice. While not a direct antiviral agent, BPC-157 research demonstrates potent systemic anti-inflammatory activity, upregulation of nitric oxide synthesis, and protection of neural tissue. Given that neuroinflammation within the trigeminal ganglion is a documented precursor to HSV-1 reactivation, BPC-157's neuroprotective and anti-inflammatory profile makes it a compound of research interest in this context. Animal model studies have demonstrated BPC-157's ability to modulate cytokine cascades and reduce oxidative stress in neural tissue. Research protocols typically explore dosing in the range of 200–500 mcg per day, administered subcutaneously or intranasally, though this remains an area of ongoing investigation.
Thymosin Alpha-1 (Tα1)
Thymosin Alpha-1 is a thymic peptide with well-documented immunomodulatory properties. It enhances T-cell maturation, increases NK cell cytotoxicity, and upregulates interferon-gamma — a cytokine directly involved in maintaining HSV latency by suppressing viral gene expression in infected neurons. Thymosin Alpha-1 has been clinically studied in the context of chronic viral infections, including hepatitis B and C, and its mechanism of action is directly relevant to herpes suppression. Research has explored doses of 1.6 mg subcutaneously twice weekly in immunocompromised contexts, with immune-support protocols varying based on individual immune status.
NAD+ Precursors: NMN and NR (Nicotinamide Mononucleotide / Nicotinamide Riboside)
Because mitochondrial ATP depletion in sensory neurons is a direct driver of HSV reactivation, restoring cellular energy metabolism is a legitimate therapeutic target. NMN (Nicotinamide Mononucleotide) and NR (Nicotinamide Riboside) are NAD+ precursors that support mitochondrial electron transport chain efficiency, upregulate SIRT1 and SIRT3 (sirtuins with antiviral regulatory functions), and reduce oxidative stress in neural tissue. Research doses range from 250–500 mg NMN daily or 300–1,000 mg NR daily, with ongoing human trials assessing systemic metabolic outcomes.
Vitamin D3 and K2
Vitamin D3 deficiency is independently associated with increased HSV reactivation frequency. Vitamin D receptors are expressed on immune cells including T-cells and macrophages, and D3 directly regulates the expression of antimicrobial peptides like cathelicidin, which has demonstrated antiviral activity against HSV. Optimizing serum 25-hydroxyvitamin D levels to the 60–80 ng/mL range (assessed via blood panel) through supplementation of 5,000–10,000 IU D3 daily combined with 100–200 mcg K2 (MK-7 form) for cofactor balance is a well-supported foundational strategy.
Lifestyle Protocol for Reducing Outbreak Frequency
No compound protocol operates effectively in isolation. The following lifestyle factors directly modulate the three root biological drivers of HSV reactivation:
- Sleep optimization: 7–9 hours of consolidated sleep per night is non-negotiable. Sleep deprivation acutely elevates cortisol, suppresses T-cell function, and drives systemic inflammation — all three reactivation triggers simultaneously.
- Blood glucose regulation: A low-glycemic, whole-food dietary approach reduces the insulin resistance that impairs immune surveillance. Eliminating refined carbohydrates and ultra-processed foods is a foundational step.
- Arginine-to-Lysine dietary ratio: Minimize high-arginine foods (peanuts, almonds, chocolate, oats, seeds) and prioritize high-lysine foods (lean meats, fish, eggs, legumes) during high-stress or high-vulnerability periods.
- Stress and cortisol management: Chronic psychological stress is the single most consistent reactivation trigger documented in human studies. Measurable cortisol reduction through structured breathwork, HRV training, and adaptogenic compounds (ashwagandha, rhodiola) supports immune homeostasis.
- UV protection: Ultraviolet radiation is a well-established HSV-1 reactivation trigger, particularly for lip-area outbreaks. SPF-rated lip balm applied daily during sun exposure reduces this specific trigger significantly.
Frequently Asked Questions
Why do cold sores keep coming back even when taking antiviral medication?
Antiviral medications like acyclovir and valacyclovir inhibit active viral replication but cannot reach or eliminate the latent viral DNA residing in sensory nerve ganglia. When the underlying biological triggers — systemic inflammation, insulin resistance, and mitochondrial energy depletion — are not corrected, the immune environment that permits reactivation remains in place, and outbreaks continue regardless of medication use.
Is there a cure for herpes simplex virus?
There is currently no cure for HSV-1 or HSV-2. The virus establishes permanent latency within sensory neurons, and any approach capable of fully eliminating it would necessarily involve destroying the infected nerve cells themselves. The goal of evidence-based management is suppressing reactivation frequency to near-zero by addressing the biological conditions that permit reactivation, not pursuing an impossible eradication.
What triggers a cold sore outbreak?
The most consistently documented triggers include psychological stress (which elevates cortisol and suppresses immune function), sleep deprivation, UV radiation exposure, febrile illness, physical trauma to the lip area, poor blood sugar regulation, and nutritional deficiencies — particularly in zinc, vitamin D, and lysine. These triggers converge on the same three pathways: inflammation, metabolic dysfunction, and cellular energy failure.
Does L-Lysine actually work for cold sores?
L-Lysine is one of the most clinically validated natural interventions for HSV-1 frequency reduction. Its primary mechanism involves competitive inhibition of L-arginine, which the herpes virus requires for replication. Multiple clinical trials support daily supplementation in the range of 1,000–3,000 mg for prophylactic use, with higher acute doses explored during prodrome periods. It is most effective when combined with dietary arginine restriction.
What is BPC-157 and how might it relate to herpes outbreaks?
BPC-157 is a synthetic research peptide derived from a naturally occurring gastric protective protein. Its relevance to herpes management is indirect but mechanistically coherent: BPC-157 demonstrates potent systemic anti-inflammatory and neuroprotective activity in animal models, targeting cytokine pathways and oxidative stress in neural tissue — both of which are documented contributors to HSV reactivation in the trigeminal ganglion. It is not an antiviral agent, but its ability to address one of the three core drivers of reactivation makes it a subject of ongoing research interest.
Key Takeaways
- HSV-
🛒 Shop Featured Peptides
Peptides mentioned in this article are available from Mile High Compounds. Use our referral link to support independent peptide research.
These are third-party products. This is not medical advice. Consult your physician before use. Affiliate links support this site.